Researchers Use Light to Make Their Microscopic ‘Muscle’ Contract on Command
Apr 10, 2026 —
Engineers interested in creating artificial cells to deliver drugs to unhealthy parts of the body face a key challenge: for a cell-like system to move, change shape, or divide, it needs a way to generate force on command.
Biological cells rely on adenosine triphosphate (ATP) to move muscles, transport substances across membranes, and perform other functions. Many cellular machines couple ATP hydrolysis (a process where chemical energy stored in ATP is released) directly to motion.
But some single-celled organisms called ciliates use a different strategy. A pulse of calcium triggers an ultrafast contraction, and ATP is used afterward to pump calcium back into storage and reset the system.
In a Nature Communications study led by Georgia Tech, researchers learned how to use a similar mechanism to control the movements of artificial protein networks without relying on ATP-powered motor proteins. Instead, they used calcium as a trigger to make the networks contract or relax.
“If engineers want synthetic cells that can do cell-like things, they need a way to generate force on command,” said Saad Bhamla, a co-author and an associate professor in Georgia Tech’s School of Chemical and Biomolecular Engineering. “Cells have to move, change shape, and divide. We’re trying to build a controllable engine from simple parts.”
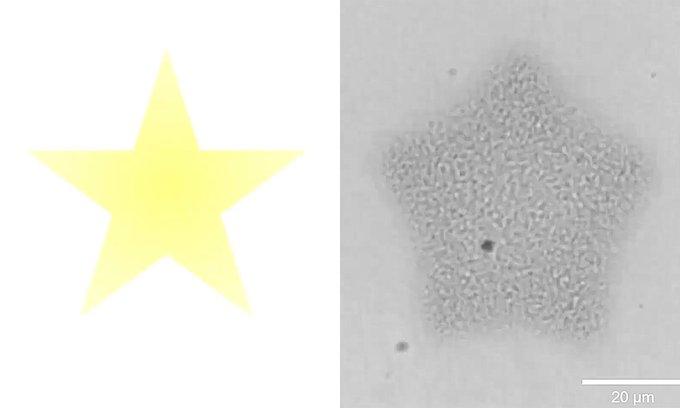
In the National Science Foundation-funded study, the team produced and purified Tetrahymena thermophila calcium-binding protein 2 (Tcb2), which is found in ciliates. The protein forms a fibrous network and contracts when exposed to calcium. The researchers reconstituted Tcb2 protein networks in the lab and then used a light-sensitive calcium chelator (a “cage” molecule that holds the calcium until illuminated) to control when and where calcium was released.
They projected light patterns of stars and circles to prompt the network to assemble and contract in matching shapes. Then, to continuously “recharge” the system, the multi-university team pulsed the light on the protein networks, repeatedly releasing calcium and driving cycles of assembly and contraction.
Jason Maderer
Director of Communications | College of Engineering





