Quiroz Peeling Back the Layers of Skin Formation
Aug 03, 2021 — Atlanta, GA

Alexa Avecilla and Felipe Garcia Quiroz
Our skin is a super organ: protective, large, flexible, powerful, self-healing, essential, totally exposed, yet still enigmatic. Felipe Garcia Quiroz, assistant professor in the Wallace H. Coulter Department of Biomedical Engineering at Georgia Tech and Emory University, works at unraveling mysteries embedded in the skin.
As a postdoctoral fellow at Rockefeller University, his research led to the discovery that liquid-liquid phase separation – LLPS, a new and hot concept in cell biology – drives formation of the skin barrier. He was lead author of a 2020 paper in the journal Science describing the research.
Now principal investigator in his own lab, Quiroz is senior author of a review article discussing the implications of his discovery on the field of skin research. The work was published recently in JID Innovations, a journal from the Society for Investigative Dermatology.
“The Science article provided us with a remarkable platform to communicate our biomolecular tools and discoveries to the broad research community,” said Quiroz, also a researcher in the Petit Institute for Bioengineering and Bioscience at Georgia Tech. The new article “presents a stimulating outlook on how the discovery of LLPS in skin, and our new bioengineered tools, will shape the future of research in skin biology and skin barrier diseases.”
Both articles focus on work by Quiroz and his colleagues illuminating the process of living skin cells migrating outward toward the body surface. There, facing exposure to the grueling external environment, the skin cells transform: They lose their nuclei and other organelles and, eventually, replace sloughed-off cells to replenish our skin barrier — a network of flat, tightly sealed cells called corneocytes.
The researchers’ focus has been on dense protein deposits that form in the skin cells before they become corneocytes — resembling droplets of vinegar in oil. This is phase separation in action, when liquids of mismatched properties come together.
These protein deposits are called keratohyalin granules, or KGs, and they resemble other membraneless organelles in cells, because they are not bound by lipid membranes. An absence of KGs is common in skin barrier disorders. Despite this strong association with human disease, the function of KGs was unknown.
Quiroz and his colleagues developed transgenic mice with a fluorescent phase separation sensor, which helped them identify the key role KGs play in skin. Their work demonstrated that intrinsically disordered proteins of the skin program the formation and properties of KGs through a vinegar-in-oil type of phase separation.
The main protein involved, filaggrin — or FLG — is often mutated in skin barrier disorders. Basically, when FLG is faulty and phase separation doesn’t happen, it opens the door to diseases of the skin barrier. The team’s research was first to establish a role for LLPS in a human tissue.
Based on the work reported in Science, Quiroz was invited by the Society for Investigative Dermatology to offer some perspective for JID Innovations. He enlisted Alexa Avecilla, a Ph.D. student in his lab, as lead author.
“Our goal in this review article is to discuss the current progress in this nascent area for skin research,” Avecilla said. “Specifically, how the discovery of liquid-liquid phase separation in skin-residing, membraneless organelles will influence understanding of the complex processes of skin barrier formation.”
The focus this time is broader than a research article; it’s more about perspective, Quiroz said, but it also works as a companion piece to the study published in Science. And it gives Quiroz and his team an opportunity to share their latest research directly with the people who ultimately will use it on the front lines of clinical care.
“We’re communicating here with the scientists and clinicians who are always thinking about diseases of the skin, the dermatologists and skin biologists,” Quiroz said. “And that will have an impact on the eventual translation of our LLPS-inspired ideas.”
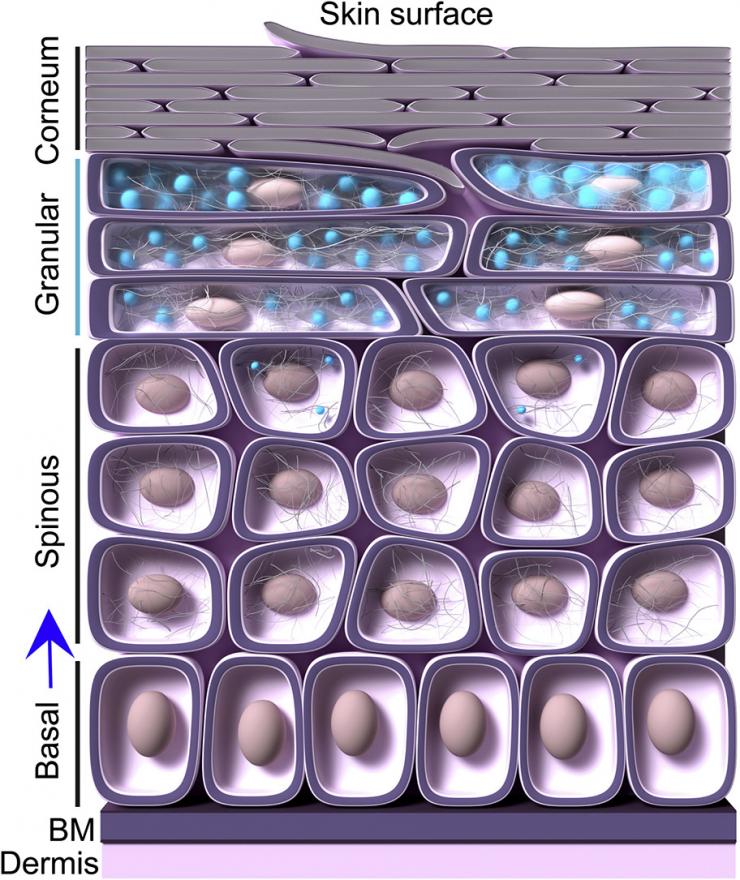
Upon detachment from the basement membrane (BM), skin cells begin their migration toward the corneum and the skin surface. In the spinous layer these cells acquire keratin filaments. Moving outward, the cells upregulate a protein called filaggrin (FLG), which triggers the assembly of keratohyalin granules (KGs) through liquid-liquid phase separation. These KGs – the blue blobs – become prominently visible in the granular layer, where FLG levels are highest. KGs grow in number and volume in the granular layer, crowding cell cytoplasm. As they move into the corneum, the cells disassemble, losing KGs, nuclei, and other organelles, and becoming flattened, dead corneocytes filled only with keratin filaments. These dead cells form the protective outer layer of our bodies, until they slough off, making room for the next wave. (Illustration Courtesy: Felipe Quiroz)
Communications officer




