Pardue Brings Clarity in Pilot Study
Mar 24, 2020 — Atlanta, GA
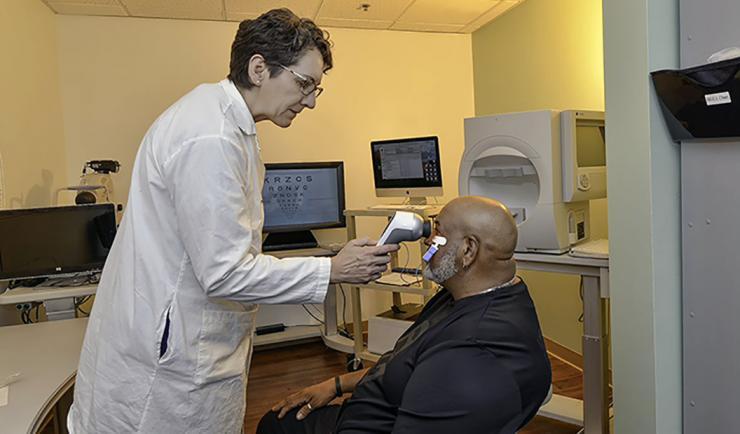
Machelle Pardue performs an electroretinogram on Army Veteran Michael Brooks, who participated in her study. (Photo by Joey Rodgers for VA)
Anyone who has diabetes can develop diabetic retinopathy, which is caused by damage to the nerve cells and blood vessels of the light-sensitive tissue in the back of the eye—the retina.
A potentially devastating eye disease, diabetic retinopathy is a leading cause of blindness among working-age Americans.
“This means vision loss is disrupted when these people are at the peak of their wage-earning potential,” says Machelle Pardue, associate director of the Center for Visual and Neurocognitive Rehabilitation at the Atlanta VA Medical Center, professor in the Wallace H. Coulter Department of Biomedical Engineering at Georgia Tech and Emory University, and researcher in the Petit Institute for Bioengineering and Bioscience at Georgia Tech.
Pardue set out to learn more about how diabetic retinopathy develops and to determine the first signs of retinal problems, with an eye on spotting early defects in the retina and treating them right away. She led a pilot study that involved the use of a hand-held portable electroretinogram (ERG) device to detect pre-clinical, or early stage, diabetic retinopathy and a neuroprotective drug to treat patients who are showing signs of the condition.
An ERG records the electrical activity of light-sensitive cells in the eyes in response to a flash of light. The response, known as the oscillatory potential, is a characteristic waveform that can be measured to determine changes in size or timing.
The drug, levodopa (commercial name Sinemet), is considered the most potent medication for treating Parkinson’s disease but isn’t approved by the U.S. Food and Drug Administration for diabetic retinopathy. Levodopa in the brain leads to the formation of dopamine, a chemical that transmits information between nerve cells and helps regulate movement, attention, learning, and emotional responses. Animal studies have suggested that a lack of dopamine may be behind early functional deficits in patients with diabetic retinopathy.
In her study, Pardue and her team found that the portable device, called the RETeval, has the sensitivity to detect early retinal dysfunction in diabetic patients prior to “clinically recognized vascular changes,” which are retina damage caused by abnormal blood flow.
“These data demonstrate the potential to use this approach as a new screening method for diabetic retinopathy that would detect retinal defects much earlier than the currently used fundus photography,” the researchers write. That procedure calls for photographing the rear of the eye, which is also known as the fundus, and recording activity in the retina.
The researchers also determined that treatment with levodopa can restore inner retinal function to normal levels within two weeks and can continue to provide benefit for at least two weeks after the treatment is stopped.
The results appeared online in February 2020 in the journal Diabetes, published by the American Diabetes Association.
Early Detection Critical
Pardue says this is the first time the RETeval has been tested in combination with levodopa for patients with pre-clinical diabetic retinopathy. It’s critical to detect the condition in its early stages, which is in the first five years after a patient is diagnosed with diabetes, and then to treat it right away, she notes. The “rule of thumb,” she says, is most people don’t get vascular changes until they’ve had diabetes for about 15 years.
“Let’s say we’re able to detect diabetic changes in the eye within the first five years,” says Pardue, who is also a professor at the Georgia Institute of Technology and Emory University, both in Atlanta. “That’s still around 10 years before it’s clinically diagnosed. That’s a big difference. When you’re at that late stage of the disease, a lot of drugs, no matter how good they are, just aren’t going to be able to treat it effectively because the disease has progressed too far. So there are sweet spots where diseases are reversible and treatable. In situations where you pass that line, it’s really hard to bring things back or even stop the disease because a cascade of events are going to lead to it progressing. To put the brakes on at that stage is really difficult.”
Diabetic retinopathy, which usually affects both eyes, is the most common diabetic eye disease. At first, the disease may cause no symptoms or only mild vision problems. But it can worsen and lead to vision loss over time. The longer one has diabetes, the greater the risk of developing diabetic retinopathy.
In some people with the disease, blood vessels may swell and leak fluid. In others, normal new blood vessels grow on the surface of the retina.
The RETeval, made by Maryland-based LKC Technologies, is the first completely portable, handheld, full-field flash ERG that measures the electrical response of the entire retina, the company says. The device is easy to transport and store. It includes a pupil tracker, eliminating the need for dilating drops, and can adjust the strength of the flash based on the pupil size. The RETeval is also used with a cheek electrode, which is a sticker that’s placed on the skin just under the eye.
Other small hand-held ERG devices are available, but they don’t include the pupil tracker. “The fact that this one is really small and portable, as well as having the pupil tracker, makes it very unique,” Pardue says, noting that she has no financial ties to LKC Technologies. “Thus, this device enables an ERG to be recorded anywhere without eye drops or an electrode touching the eye.”
The RETeval, she adds, has been used by clinicians at the clinical stage of diabetic retinopathy. “What’s new about our study is that we’re not trying to detect diabetic retinopathy at that stage, which I argue is late stage,” she says. “We’re trying to detect it much earlier, when we can use drugs that are actually going to prevent the progression of the disease.”
Convenient Tool
The electroretinogram can be thought of an electrocardiogram, or an EKG for the eye. After electrodes are placed on or near the eye, electrical activity of the retina is recorded in response to a flash of light. Over the years, ERGs have been carried out in ophthalmology clinics to measure retinal function for clinical diagnostics and monitoring. But the systems are large and cumbersome and require an expert to conduct the test and analyze the data, Pardue says.
The typical ERG, she explains, requires dilation drops to enlarge the pupils, dark-adaptation of up to 30 minutes to make the retina very sensitive to light, and an electrode placed on the cornea after numbing drops are applied. All of these characteristics make current ERG systems not conducive to a screening test for the disease.
“When I think of a screening test, I think of something that’s going to be very easy to perform and that will produce a result in a very short amount of time,” she says. “Instantaneously would be great. For example, blood pressure cups are a successful screening tool—the ones you see in the pharmacy where you sit down and put your arm in, and a few seconds later you have your blood pressure numbers. The ERG as it’s now performed is not conducive to that. However, with the adoption of the RETeval, we may be close.
“Also, ERGs aren’t accessible everywhere,” she adds. “They’re really only found in large university-based ophthalmology clinics because those sites have people with expertise to analyze that information. A good screening tool could be in a pharmacy or in a primary care clinic, where a technician could run it quickly and easily and have a simple output that will tell you this patient is normal or an issue needs to be looked at more closely.”
Pardue’s study included 43 Veterans at the Atlanta VA ages 37 to 71. Fifteen of the Veterans were non-diabetics who didn’t take levodopa in a control group. Pardue and her team used the RETeval with those patients to record a normal electrical activity range in the retina that was based on the oscillatory potential (OP).
The other 28 Veterans had diabetes and retinal deficits spotted by the RETeval, based on readings that were outside of the control group’s normal range. They were randomized to receive low- or high-dose levodopa, which was given orally, for the first two weeks of the study. In a two-week “washout” period that ensued, the researchers checked via ERGs for any sustained effects from the drug. Those patients had four ERGs over the four-week period: at baseline, day one, week two, and week four.
Clinical Trial Planned
The ERG consisted of sitting in the dark for 10 minutes, with an adhesive sensor strip placed on the skin near the eye. The researchers positioned the RETeval in front of the right eye and left eye separately, applying a series of flashes and flickering lights. Both eyes were evaluated in less than five minutes for most patients.
The research team found both doses of levodopa to be equally effective. But to avoid potential side effects, Pardue favors the lowest dose possible because patients with diabetic retinopathy could be taking the drug for many years. The possible side effects of levodopa are many, ranging from anxiety and agitation to numbness and dizziness.
“These exciting data suggest that OP delays are very sensitive to diabetic changes and that reduced dopamine may be partially responsible for the early inner retinal dysfunction,” the researchers write. “After two weeks of treatment, OP delays were restored to control values that persisted even after a two-week washout period. In conclusion, … OP delays could serve as an earlier detection strategy for diabetic retinopathy that could be treated with levodopa, potentially limiting later-stage vision loss.”
Army Veteran Michael Brooks, who was diagnosed with diabetes about five years ago, participated in the treatment group. He says his vision was a little stronger after he took the levodopa but notes the improvement may have been due to the corrective laser eye surgery he had a few years ago. That procedure changes the shape of the cornea, the clear covering in front of the eye, to improve vision and reduce a patient’s need for glasses or contact lenses.
“What I gather from this study is that it was an experiment to prevent diabetic retinopathy and to identify any existing problems in the eye,” he said. “I was all on board with that. I’m a fire paramedic, so tests and research interest me. Anything that would potentially tell me I’m a candidate for having serious eye problems down the road, I’m on board.”
Pardue isn’t surprised that Brooks didn’t recognize major visual changes after taking levodopa.
“The visual system is extremely good at compensating for small defects, which is the reason it’s hard for a patient to detect early changes,” she says. “The electrical defect we can detect with the ERG is identifying a change before the patient is aware of an issue. We are thus preserving vision hopefully so the patient never has any visual loss associated with diabetic retinopathy.”
Pardue and her team plan to do a larger clinical trial to see if they can get the same benefits from levodopa when diabetic patients are treated for at least six months. She estimates up to 200 Veterans may be involved in the multi-site trial. Brooks says he’ll participate in the trial if it doesn’t interfere with an ongoing diabetes study he’s involved in with Kaiser Permanente.
“We want to know if timing is important,” she says. “Can the patients be treated later in the progression of the disease and still get benefit? We also want to move forward with developing the ERG test for screening by determining more parameters about the patients who seem to have ERG delays. Do ERG delays correlate with such factors as blood glucose levels, body mass index, and duration of diabetes? Our ultimate goal is to move this to primary care clinics, where diabetic retinopathy could be detected much earlier, followed by levodopa treatments, to prevent vision loss from occurring in later stages of the disease.”
This story was written by Mike Richman, VA Communications
Jerry Grillo
Communications Officer II
Parker H. Petit Institute for
Bioengineering and Bioscience




