Genetics and Cancer: Research Offers New Insights On Risks, Onset, Progression
Feb 18, 2020 — Atlanta, GA
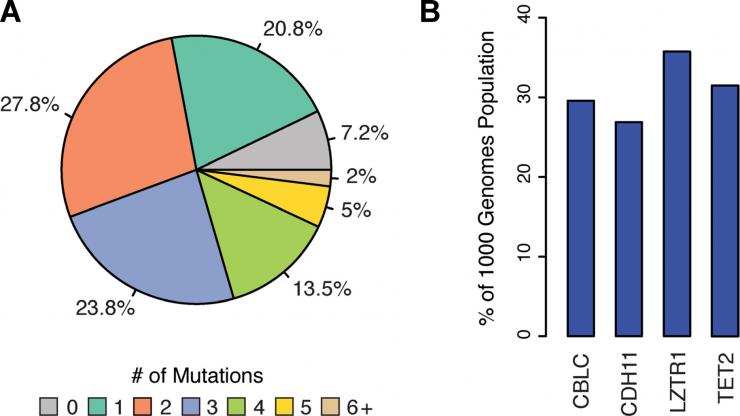
(A) Pie chart depicting the percent of the 1KGP containing deleterious cancer associated mutations in at least one tumor suppressing gene (TSG.) (B) Four TSGs most frequent
A new study by researchers in the School of Biological Sciences raises new questions about a decades-old, award-winning theory regarding how many genetic mutations are necessary for cancer to develop in human cells.
That theory, called the Knudson Hypothesis, argued that two mutations in the type of genes that suppress tumors are needed to lead to changes that could cause cancer. However, John McDonald, a School of Biological Sciences professor and the director of Georgia Tech’s Integrated Cancer Research Center, says the research, published in Oncotarget, “shows, for the first time, that nearly all normal healthy individuals carry at least one potentially cancer-causing tumor suppressor gene mutation. The implication is that a majority of the human population is, to a greater or lesser extent, predisposed to develop cancer.”
McDonald and his fellow researchers — Evan A. Clayton, Shareef Khalid, Dongjo Ban, Lu Wang and Professor I. King Jordan, all of Georgia Tech — relied on several databases, including the Catalogue of Somatic Mutations in Cancer (COSMIC), the world’s largest database of mutations associated with cancer onset and progression. The scientists combined that database with the One Thousand Genomes Project (1KGP), which lists genetic variants present, in 2,504 normal, healthy individuals. That list reflects the diversity of racial and ethnic groups randomly selected from 26 human populations around the world.
The Knudson Hypothesis, developed in 1971 by Alfred Knudson, helped physicians identify cancer-related genes, and won Knudson the prestigious Albert Lasker Award in Clinical Medical Research in 1998.
“Inconsistent with the Knudson two-hit hypothesis, we present evidence that acquisition of a second cancer-causing tumor suppressor mutation is not necessary to drive cancer onset/progression. Rather, we present evidence that, in many cases, individuals with only a single cancer-causing tumor suppressor mutation develop cancer,” McDonald says. “In these individuals, we show that the mutant gene is significantly overexpressed relative to the normal gene, thus overriding the influence of the non-mutated gene and driving cancer onset and development. Thus, in many individuals, a change in gene expression, rather than a ‘second mutational hit,’ is responsible for the cancer.”
All humans have two copies of every gene, each copy supplied by a biological parent. Two classes of genes are believed to cause the onset and progression of cancer. Oncogenes are genes that can drive cancer after a single mutation in one of the copies of the gene. In other words, those mutations are said to be “dominant” with respect to their ability to result in cancer.
Unlike oncogenes, a second class of cancer-driving genes, called tumor suppressor genes, are considered to be recessive. Knudson’s hypothesis took the point of view that mutations in each of the two copies of the tumor suppressor genes had to happen in order to drive cancer development. McDonald says the hypothesis is still widely held in 2020 by the cancer community.
“We believe that our findings are of major significance and call into question many of the assumptions underlying current methods to diagnose and treat cancer based on genomic profiling,” McDonald says.
This research was supported by the Ovarian Cancer Institute (Atlanta), Northside Hospital (Atlanta), the Deborah Nash Endowment Fund, and National Institute of Health Bioinformatics Training Grant: CRP 10-2012-03.

Renay San Miguel
Communications Officer
College of Sciences
404-894-5209




