Fast, Accurate Scientific CMOS Noise Correction Advances Imaging for Fluorescence Microscopy
Jan 06, 2020 — Atlanta, GA
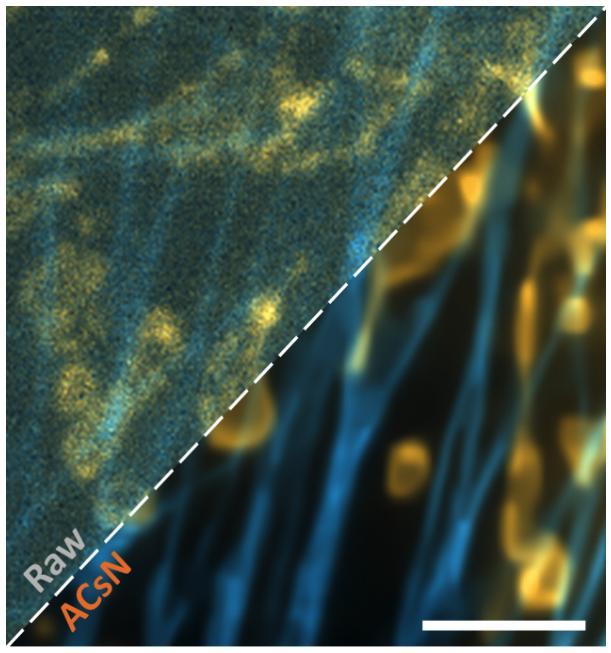
Figure: Dual-color image before and after ACsN denoising of F-actin (cyan) and mitochondria (orange) in fixed bovine pulmonary artery endothelial (BPAE) cells obtained by TIRF microscopy with an exposure time of 2 ms. Scale bar 4 um.
A fluorescence microscope is an optical microscope that uses fluorescence to study the properties of organic or inorganic substances. In today’s laboratories, these microscopes are equipped with more and more sophisticated digital cameras, which are a vital tool for scientists working in the field of bio-imaging who need to capture images to determine how living cells and tissues function. Scientific CMOS, or sCMOS, is the newest breakthrough technology that overcomes many drawbacks of past image capturing sensors.
Researcher Shu Jia, assistant professor in the Wallace H. Coulter Department of Biomedical Engineering at Georgia Tech and Emory University and a researcher in the Petit Institute for Bioengineering and Bioscience, and his team are further advancing scientific imaging with the development of a new algorithm for the automatic correction of sCMOS-related noise (ACsN) for fluorescence microscopy.
In a study published January 3, 2020 by Nature Communications, Jia’s team describes how they overcame extra noise and demonstrated significant improvements in both fluorescence microscopy images and their downstream analysis in a wide range of imaging conditions and modalities.
“The sCMOS cameras have advanced the imaging field, but they often suffer from additional noise compared to CCD sensors,” said Jia. “So we have circumvented this issue devising a content-adaptive algorithm for the correction of sCMOS-related noise in fluorescence microscopy.”
ACsN combines camera physics and layered sparse filtering to address the most relevant noise sources in a sCMOS sensor while preserving the fine details of the signal.
For example, the image acquisition of a sCMOS camera is modeled taking into account the different sources of noise present in the imaging process. Some parameters in the equation are estimated by a one-time calibration, while others are estimated directly from the captured images—including noise statistics. In addition, a layered approach to collaborative filtering is used to decompose the image in patches and sort them into 3D groups. Then, each group is jointly denoised and the new patches recombined to form the final denoised image.
The paper further illustrates how ACsN was able to improve the image quality of many common techniques such as wide-field microscopy; deconvolution and light-field microscopy; single-molecule localization microscopy; fluorescence microscopy with low-cost CMOS cameras; and selective plane illumination microscopy.
“In fluorescence microscopy, the noise introduced by the electronics of the camera is still the main limiting factor for both speed and accuracy. With ACsN, we have shown that it is possible to push such limits further without loss of information,” said Biagio Mandracchia, postdoctoral fellow in Jia’s group who led the research. “This has many implications, especially in fields like diagnostics or neural imaging of freely-moving animals, where cumbersome high-end cameras cannot be adopted.”
This research was supported by the National Institutes of Health grant R35GM124846, and the National Science Foundation grants CBET1604565 and EFMA1830941. This research project was supported in part by the Emory University Integrated Cellular Imaging Microscopy Core and by PHS Grant UL1TR000454 from the Clinical and Translational Science Award Program, National Institutes of Health, and National Center for Advancing Translational Sciences. We wish to acknowledge the core facilities at the Parker H. Petit Institute for Bioengineering and Bioscience at the Georgia Institute of Technology for the use of their shared equipment, services and expertize. T. Urner is supported by the National Science Foundation Graduate Fellowship. We thank Drs. A. Marcus and R. A. Kahn’s laboratories at Emory University for providing live-cell imaging data with lattice light-sheet microscopy.
CITATION: Biagio Mandracchia, Xuanwen Hua, Changliang Guo, Jeonghwan Son, Tara Urner and Shu Jia. Fast and accurate sCMOS noise correction for fluorescence microscopy. Nature Communications 11, 94 (2020) doi:10.1038/s41467-019-13841-8
Media Contact:
Communications Manager
Wallace H. Coulter Department of Biomedical Engineering
Georgia Institute of Technology

Pictured left to right: Biagio Mandracchia, postdoctoral fellow in Jia’s group who led the research, and Shu Jia, assistant professor in the Wallace H. Coulter Department of Biomedical Engineering.
Walter Rich




